Table of Contents
ToggleTHIS TRIAL HAS ENDED.
Please refer to the Clinical Trials page for current trials.
Intrauterine adhesions, also known as IUAs or Asherman’s Syndrome, are one of the most undiagnosed conditions among women in the United States. The adhesions are caused by damage to the lining of the uterus. The condition can lead to fertility issues and medical problems.
Fortunately, there is a new clinical trial on fibroids that is demonstrating the effectiveness of an innovative way to prevent the formation of these dangerous adhesions. The Rejoni clinical trial is led by physicians and features a gel-like material (Juveena™ Hydrogel) that may be effective at preventing the formation of intrauterine adhesions after surgery.
At Arizona Gynecology Consultants, we are committed to providing comprehensive healthcare for women using clinically proven techniques. Some of our clients may benefit from participating in this clinical trial on intrauterine adhesion.
What Are Intrauterine Adhesions?
The interior of the uterus is lined with a type of tissue called endometrium. During pregnancy, the embryo implants in this layer, but if no pregnancy occurs, the top layer of the endometrium is shed during menstruation. Any damage that occurs to the lining of the uterus can lead to the formation of scar tissue between the inner walls of the uterus.
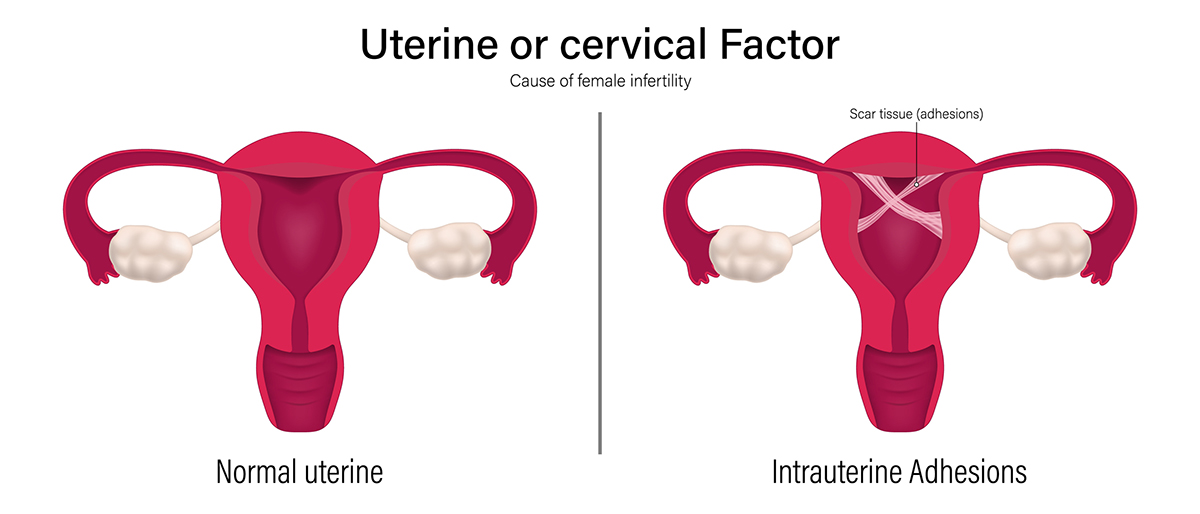
Thin bands of scar tissue may form if the scarring is mild. Thick bands may form when the scarring is more severe. In either case, partial or total occlusion (blockage) of the inside of the uterus can occur.
The resulting conditions, commonly referred to as Asherman’s Syndrome, can develop after a uterine procedure like a dilation and curettage or hysteroscopy. Asherman’s Syndrome can cause serious fertility issues that require gynecology services in Arizona.
Primary adhesions can appear after pregnancy loss or surgery to remove fibroids. Secondary adhesions can happen after surgery to remove them, resulting in the adhesions returning. The adhesions may or may not cause noticeable symptoms.
How Does the Clinical Study Work?
Rejoni, Inc., a company based in Bedford, MA, is heading the study that tests whether a new hydrogel can prevent the formation of intrauterine adhesions. Any patient who is planning to undergo uterine surgery (either to disrupt existing adhesions through a procedure known as adhesiolysis or to remove fibroids) may be a candidate for the study.

Eligible participants will be randomly assigned to one of two groups: the treatment group, which will receive the hydrogel immediately after uterine surgery, or the control group, which will not receive the hydrogel. Two participants will be assigned to the treatment group for every participant in the control group. If you’re interested in this research trial, please schedule an appointment with Arizona Gynecology Consultants to determine if you qualify as a candidate.
Am I a Candidate For This Clinical Study?
If you have a history of intrauterine adhesions, you may be a great candidate for the Rejoni clinical study. Candidates may be able to prevent the recurrence of intrauterine adhesions by participating in the study. Women who have a history of being diagnosed with these types of adhesions may be good candidates for the clinical study.
If you are at least 18 years old and have received a diagnosis indicating that you require fibroid removal or adhesiolysis, you may qualify to participate in the Rejoni clinical trial or any similar clinical trial. There are no expenses associated with participating in this research. By joining this study, you can contribute to advancements in medical knowledge and potentially benefit from cutting-edge treatment options.

You will be provided with detailed information about the study procedures beforehand and questions you have will be thoroughly answered. Your participation could make a significant difference in the lives of others affected by similar conditions.
What Causes Intrauterine Adhesions?
Intrauterine adhesions can form after a surgical procedure involving the cavity of the uterus. Dilation and curettage (often used to diagnose and treat conditions) is an outpatient procedure that involves stretching the opening to the uterus and selectively removing parts of the uterus. The surgical procedures can result in intrauterine adhesions.
Dilation and curettage are often performed to control uterine bleeding and to address pregnancy complications. Less common causes of intrauterine adhesions include endometritis, surgical removal of fibroids, endometrial ablation, and cesarean sections.
What Are the Symptoms of Intrauterine Adhesions?
Intrauterine adhesions may cause significant symptoms or no symptoms at all.
Common symptoms include:
- Menstrual abnormalities
- Light or infrequent periods
- Inability to achieve pregnancy
- Recurrent miscarriages
- Birth complications
- Abnormal implantation of the placenta
- Pelvic pain
- Painful menstrual periods
These could be the symptoms of Asherman’s Syndrome or another medical problem that requires treatment at our Arizona gynecology center. We offer consultations for any of our exams and use minimally invasive treatments when possible.
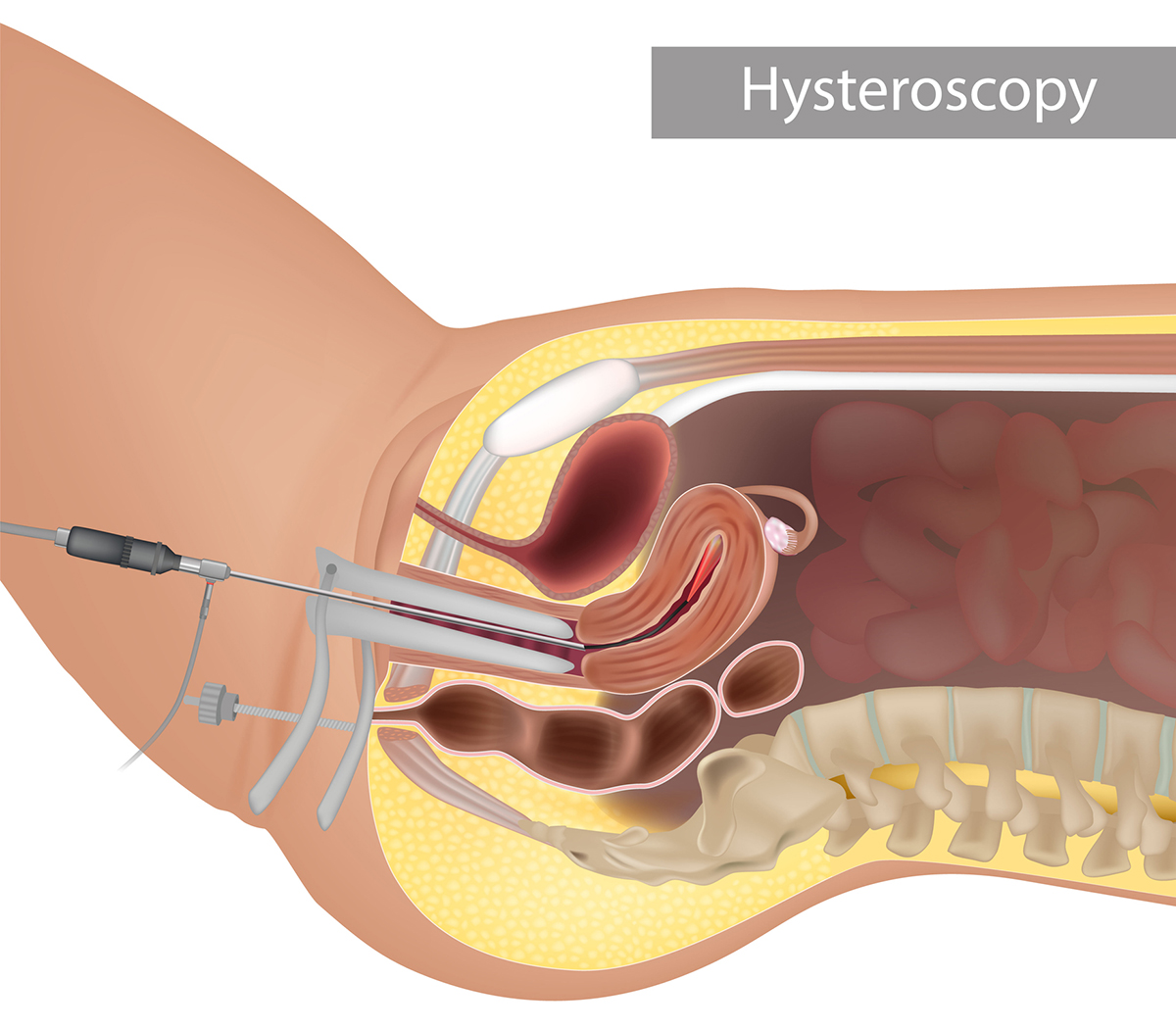
How Are Intrauterine Adhesions Diagnosed?
Asherman Syndrome is often diagnosed by hysterosalpingogram, hysteroscopy, or saline sonohysterogram. Hysteroscopy is a highly accurate method for diagnosing intrauterine adhesions. It uses a lighted telescopic instrument to allow the doctor to see inside the uterus. The procedure can be performed in the operating room or a medical office.
Imaging procedures that use a special dye can view the shape of the inside of the uterus. A saline sonohysterogram uses a saltwater solution to expand the uterus so a sonogram machine can see the uterine cavity. Imaging can discover spaces where the fluid does not freely flow. While these procedures may not require anesthesia, anti-inflammatory medications may be prescribed to decrease post-procedure cramping.
Who Is at Risk For Intrauterine Adhesions?
Several factors can increase the risk of developing intrauterine adhesions. Women who have undergone uterine surgery following miscarriage, abortion, or delivery are at risk due to the potential for scarring. Certain surgical procedures like myomectomy for fibroid removal or the presence of uterine fibroids can lead to scarring.
Postpartum infections, endometritis, or genital tuberculosis can also increase the risk. When an intrauterine device (IUD) is not inserted correctly or left in place for extended periods, the device can damage the uterine lining and increase the risk of adhesions. Genetics is another leading factor, as some women may be more predisposed to developing scar tissue in the uterus.
Women may be at higher risk of developing intrauterine adhesions if they:
- Have experienced complications related to cesarean sections
- Have experienced repeated miscarriages
- Are older
- Have a condition that causes prolonged uterine bleeding, such as pelvic inflammatory disease.
Understanding these risk factors is crucial for identifying women who may be at higher risk of intrauterine adhesions and providing appropriate preventive measures or treatment.
Common Treatments For Intrauterine Adhesions
Physicians often recommend surgical treatments to address intrauterine adhesions because the scarring can lead to medical complications and fertility issues. Once the adhesions have been identified and diagnosed, your physician may use a special operating hysteroscope to cut and remove the scar tissue.
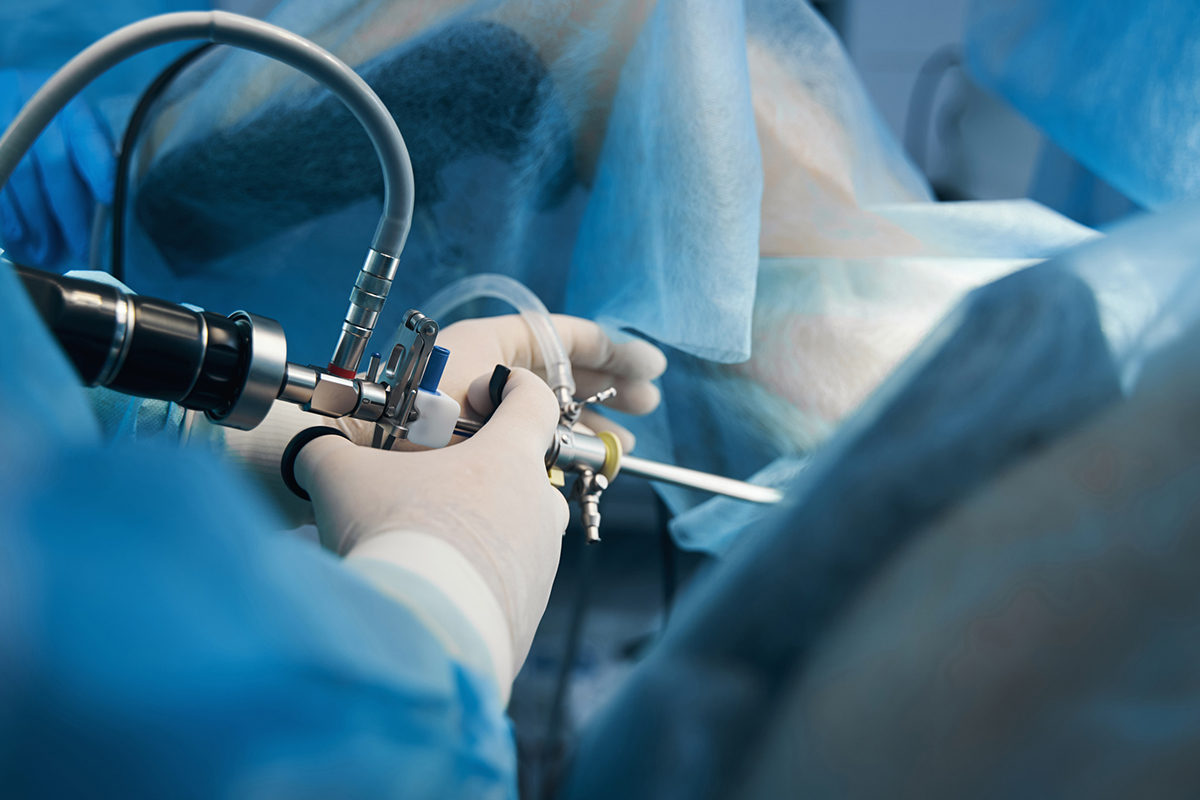
This procedure is frequently performed under anesthesia. After the adhesions have been removed, a special device may be placed to separate the walls of the uterus and decrease the chances that the adhesions will return.
To further reduce the risk of adhesions returning, your physician may prescribe hormonal treatment, nonsteroidal anti-inflammatory drugs, or antibiotics. The treatments are highly effective, but some women experience post-surgery lesions. The ongoing clinical trial on intrauterine adhesion aims to prevent the return of these types of adhesions.
Are There Risks Associated With Treating Intrauterine Adhesions?
The clinical trial may be able to prevent the return of intrauterine adhesions in some women. Any surgical procedure to remove existing adhesions can lead to absent or infrequent periods.
Women should be mindful that pregnancies after treatment could potentially be complicated by:
- Miscarriage
- Preterm labor
- Third-trimester bleeding
- Abnormal attachment of the placenta
Your women’s healthcare provider will explain how surgical removal of adhesions can affect future pregnancies and births. Women with mild-to-moderate adhesions often experience a resumption of normal menstrual function. Severe adhesions can affect fertility in some women. The Rejoni clinical trial is an important study because the advanced gel placed after surgical removal of adhesions may prevent the return of intrauterine adhesions.
Rejoini Clinical Trial FAQs
If you believe you are a good candidate for this or another ongoing clinical trial, please let someone with Arizona Gynecology Consultants know. We can determine your candidacy and help you start the process of undergoing treatment.
A surgical procedure known as adhesiolysis treats intrauterine adhesions caused by Asherman’s syndrome. Under general anesthesia, a doctor dilates the cervix. This allows a hysteroscope to be inserted into the uterus. The surgeon identifies and removes scar tissue using a special instrument. The doctor then controls any bleeding. Patients are monitored after the procedure and given pain management if needed.
If you are 18 and older and have a history of developing intrauterine adhesions or have undergone fibroid removal or adhesiolysis, you may be a good candidate. The treatment prevents the return of adhesions following surgery. If you do not have intrauterine adhesions or a history of developing this type of scarring, you may not be a candidate for this specific clinical trial.
For every person selected as part of the control group, two women will be selected for the treatment group. These women will be given Juveena™ Hydrogel following the removal of an intrauterine adhesion. The study assesses whether the application of the gel prevents the return of adhesions in the uterus.
By participating in the Rejoni clinical trial, you may be chosen for the treatment group. The treatment gel may reduce the chances that intrauterine adhesions return. Preventing the return of adhesions could reduce the risk of fertility issues and other problems related to Asherman’s Syndrome.
Before you can sign up for this clinical trial, one of our physicians will determine if you are a good candidate. We will provide you with everything you need to participate if you are found to be a candidate. At Arizona Gynecology Consultants, we are following national clinical trials and will recommend new clinical trial opportunities as they arise.
Contact Arizona Gynecology Consultants to See If You Are a Clinical Trial Candidate
Women who participate in the Rejoni clinical trial may be able to prevent the return of intrauterine adhesions. If you have a history of intrauterine adhesions and are 18 or over, you may be a great candidate for this study.

Arizona Gynecology Consultants is continually following new and ongoing clinical studies related to women’s health. When a study offers a safe and effective potential treatment for a condition we treat, we may recommend that our patients participate in the study.
It is important to remember that studies may involve the use of a control group, meaning that participating does not guarantee that you will have access to the new treatment being tested. Participation does help grow medical science’s body of knowledge, and you may directly benefit from those advancements. To learn if you are a candidate for the Rejoni clinical trial, please contact our office today.
THIS TRIAL HAS ENDED.
Please refer to the Clinical Trials page for current trials.
Sources:
- Parashar, S., Pajai, S., & Tarang, T. (n.d.). Recent Advancement in the Management of Intrauterine Adhesions Using Stem Cell Therapy: A Review Article. Cureus, 15(8), e43553. https://doi.org/10.7759/cureus.43553
- Smikle, C., Yarrarapu, S. N. S., & Khetarpal, S. (2022). Asherman Syndrome. PubMed; StatPearls Publishing. https://www.ncbi.nlm.nih.gov/books/NBK448088/#:~:text=Asherman%20syndrome%20(intrauterine%20adhesions%20or
- Intrauterine adhesions: what are they? (n.d.). Www.reproductivefacts.org. https://www.reproductivefacts.org/news-and-publications/fact-sheets-and-infographics/intrauterine-adhesions-what-are-they/

Founder and Medical Director of ARIZONA GYNECOLOGY CONSULTANTS
Dr. Kelly Roy is a specialist in surgical gynecology and advanced laparoscopy (and hysteroscopy). She is a long-time resident of Arizona and obtained her Bachelor of Science degree in Biomedical Engineering at Arizona State University before finishing her Doctorate of Medicine at the University of Arizona in 1997.
Dr. Roy completed her residency in Obstetrics and Gynecology at the then “Banner Good Samaritan Hospital” (now Banner University Medical Center), in Phoenix Arizona in 2001.
Well known for her teaching and surgical ability, she is on the faculty at the residency program at both Banner University Medical Center and Saint Joseph’s Hospital in central Phoenix and is a Clinical Assistant Professor of Medicine at the University of Arizona College of Medicine, Phoenix Campus. Dr. Roy has taught advanced surgical techniques to medical students, residents, fellows and colleagues for over 15 years.
Dr. Roy is also a consultant to the medical device industry and has participated in the design and clinical testing of many instruments and surgical devices available on the world-wide market today.
Read More About Dr. Kelly Roy, MD | WebMD Profile | Health.USNews.com Profile | Current Obstetrics and Gynecology Reports: TFA with the Sonata System



